Temziva can be used to treat Cytokine Release Syndrome (CRS) as the one of The danger cause of Corona disease in adults and pediatric patients (2 years of age and older) with confirmed diagnosis of covid-19.
Microbial
patient information leaflet
Microbial
Temziva
Temziva contains the active substance tocilizumab, that blocks the action of a specific protein (cytokine) called interleukin-6. IL-6 activity in body causes many symptoms of autoimmune disease such as Rheumatoid Arthtitis (RA). Blocking IL-6 receptors can reduce the inflammation in your body and reduce symptoms.
Tocilizumab is used for:
Currently, Temziva has been approved by the Food and Drug Administration for the treatment of Cytokine Release Syndrome (CRS) as one of the dangers which cause corona disease in adults and pediatric patients (2 years of age and older) with confirmed diagnosis of covid-19.
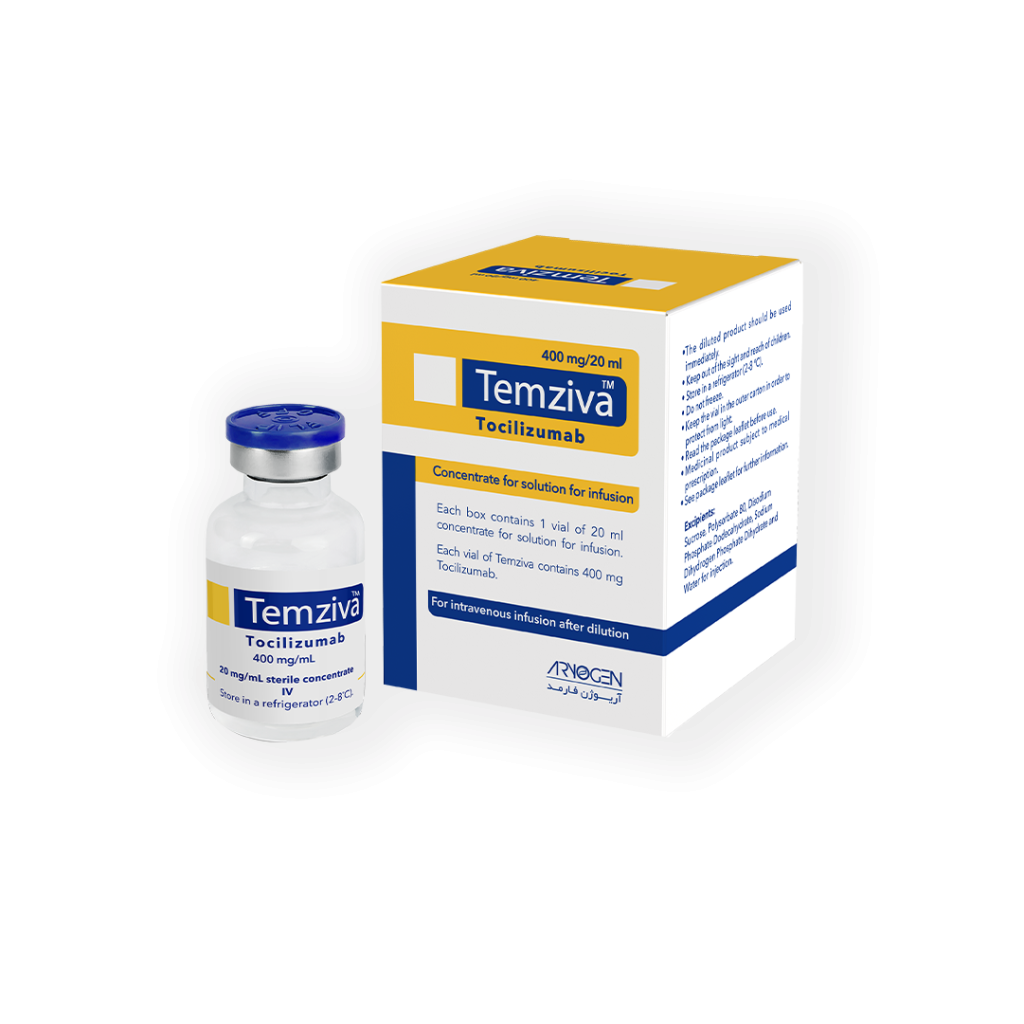
Temziva vial contains 400mg/20ml concentrate for solution for intravenous injection. The active substance is tocilizumab. Each 20ml vial contains 400mg tocilizumab (20mg/ml). Each pack contains 1 vial of concentrated solution and a patient leaflet. Temziva is infused intravenously. Drug solution should be diluted by sodium chloride 9mg/ml (0.9%) solution for injection and given through intravenous infusion for atleast over one hour.
AryoSeven
AryoSeven is produced using recombinant BHK cells, which is stored in liquid nitrogen (-185) under cell bank regulations.
Altebrel
Active Raw Material (API) of Altebrel with the name of Etanercept is being purified via several pre-purification and purification steps by chromatography methods for use in final formulation.
Zytux
Zytux™ is a biosimilar product with the generic name of Rituximab. It is a genetically engineered human-mouse chimeric monoclonal antibody produced by Chinese Hamster Ovary (CHO) cells in suspension.
AryoTrust
AryoTrust (Trastuzumab) is produced by hiring recombinant CHO cells as small factories for production of the therapeutical protein.
Stivant
Stivant is used in different types of cancers such as; metastatic cancer of colon or rectum, progressive glioblastoma, Non-Small Cell Lung Cancer, advanced or metastatic cancer of kidney, ovarian, cervical cancer and metastatic breast cancer.
Temziva
Temziva can be used to treat Cytokine Release Syndrome (CRS) as the main danger cause of Corona disease in adult patients with confirmed diagnosis of covid-19.
Arylia
Arylia contains the active substance Denosumab with a protein stnucture. Denosumab is a human monoclonal antibody (lgG2) that targets and binds with high affinity and specificity to RANKL.
VedAryo
Arylia contains the active substance Denosumab with a protein stnucture. Denosumab is a human monoclonal antibody (lgG2) that targets and binds with high affinity and specificity to RANKL.